At Vivoryon, we are focused on developing innovative small molecule-based medicines for the treatment of inflammatory and fibrotic disorders of the kidney. Driven by our passion for ground-breaking science and innovation, we strive to improve patient outcomes by changing the course of severe diseases through modulating the activity and stability of pathologically relevant proteins. Our most advanced program, varoglutamstat, a proprietary, first-in-class orally available QPCT/L inhibitor, is being evaluated to treat diabetic kidney disease.
Pipeline
Developing a pipeline of small molecule enzyme inhibitors to treat severe diseases
Many different diseases are caused and/or promoted by post-translational modification of proteins. For a number of disease indications, Vivoryon has shown that the enzymes catalyzing these reactions can successfully be targeted with small molecule enzyme inhibitors.
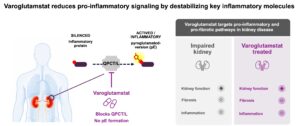
Our most advanced development program, varoglutamstat targets the glutaminyl cyclases QPCT and QPCTL. These enzymes catalyze a specific post-translational modification process known as pyroglutamate formation, which is the formation of a ring at the N-terminus of a number of different peptides and proteins. This post-translational modification changes the properties of the proteins and the modified proteins have been shown to play important roles in causing or promoting a number of different diseases.
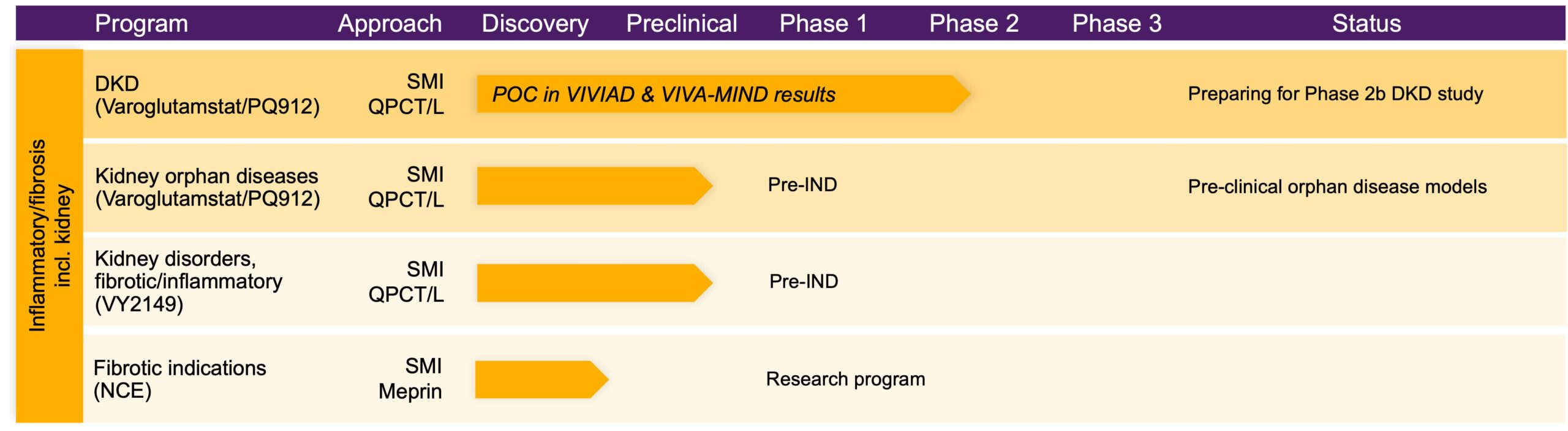
Blocking these enzymes to prevent the modifications enables an intervention early in the different pathologic pathways. We are developing a pipeline of orally administered small molecule inhibitors of glutaminyl cyclases, with our most advanced medicine in development, varoglutamstat, having demonstrated a beneficial effect on kidney function in two independent Phase 2 studies in an elderly patient population. Based on these findings we are focused on pursuing development of varoglutamstat in kidney disease.
Beyond QPCT/L inhibitors, we are assessing the potential of meprin inhibitors and also have programs to develop selected monoclonal antibodies (mAbs).
Varoglutamstat
Lead Candidate Varoglutamstat
Our lead candidate, varoglutamstat (PQ912) is a proprietary, potent (nanomolar) and selective inhibitor of human glutaminyl cyclases QPCT and QPCTL with therapeutic potential in a diverse set of indications with high unmet medical need including inflammatory/fibrotic disorders of the kidney and other organs.
Our ongoing varoglutamstat Phase 2 program is currently focused on kidney disease and has shown highly consistent, statistically significant and clinically meaningful improvement of kidney function (eGFR) versus placebo as well as an exceptional safety profile in two independent randomized double-blind placebo-controlled studies, VIVIAD* and VIVA-MIND*. Results to date highlight the unique potential of varoglutamstat to meaningfully improve kidney function. The effect of varoglutamstat on eGFR observed in both studies was greater in elderly participants with diabetes compared to elderly participants without diabetes. Based on these findings, Phase 2 development for varoglutamstat is primarily being pursued in diabetic kidney disease.
* Phase 2 studies in early Alzheimer’s disease included prospectively defined measures of kidney function as safety and other exploratory endpoints
QPCT/L inhibitors in kidney inflammation/fibrosis:
Over a decade of research and know-how supports scientific rationale
Persistent low-grade inflammation is now considered a hallmark feature of chronic kidney disease (CKD)1
There is extensive scientific proof that many inflammatory and fibrotic pathways require formation of N-terminal pyroglutamates (pE) for full activity and the pyroglutamate (pE) versions of chemokines like CCL-2 and CX3CL1 (fractalkine) have been shown to be increased in chronic kidney disease (CKD) and may contribute to a number of renal diseases2,3,4,5
CCL2 is a validated target in kidney disease
QPCT/L inhibition has been shown to improve kidney function and reduce inflammation in a glomerulonephritis CKD rat model via CCL2/CCR2 axis, using a Vivoryon compound6 and CCL2 deficiency has been demonstrated to protect against chronic renal injury in murine renovascular hypertension7 . Blocking CCL2/CCR2 signaling ameliorates diabetic nephropathy in the db/db mouse model of type 2 diabetes8. CCL2 plasma levels were significantly higher in patients with CKD compared to the control group9.
-
1 Yilmaz MI et.al., Clin Nephrology 2007
-
2 Tesch GH, Am J Renal Physiol 2008
-
3 Kehlen et.al , Biosci Rep, 2017
-
4 Cynis et.al., EMBO 2011
-
5 Cynis et.al, Intl Jour. of Exp. Pathology 2013
-
6 Kanemitsu et.al., Naunyn-Schmiedeberg’s Arch. of Pharmac., 2021
-
7 Kashyap et al. Sci Rep. 2018
-
8 Soek et al. Nephrol Dial Transplant, 2013
-
9 Vianna et al., Pediatr Nephrol, 2013
Clinical proof of concept data:
Varoglutamstat improved kidney function in completed Phase 2 VIVIAD and VIVA-MIND studies
Based on the known anti-inflammatory activity of varoglutamstat, the protocol for the Phase 2 VIVIAD and VIVA-MIND studies in early AD, which were completed in 2024, included the investigation of kidney function and measurement of biomarkers of kidney inflammation and fibrosis to explore the role of QPCT/L inhibition on kidney function. Although patients in these studies were selected for their AD status and not for their kidney function level, many of them had reduced kidney function due to age and or comorbidities. The two independent Phase 2 clinical studies demonstrated a statistically significant improvement in kidney function (as measured by an increase in eGFR) under treatment with Vivoryon’s lead candidate, varoglutamstat. The treatment effect, in particular in patients with diabetes, was unprecedentedly large and was shown to be sustainable over a period of two years.